 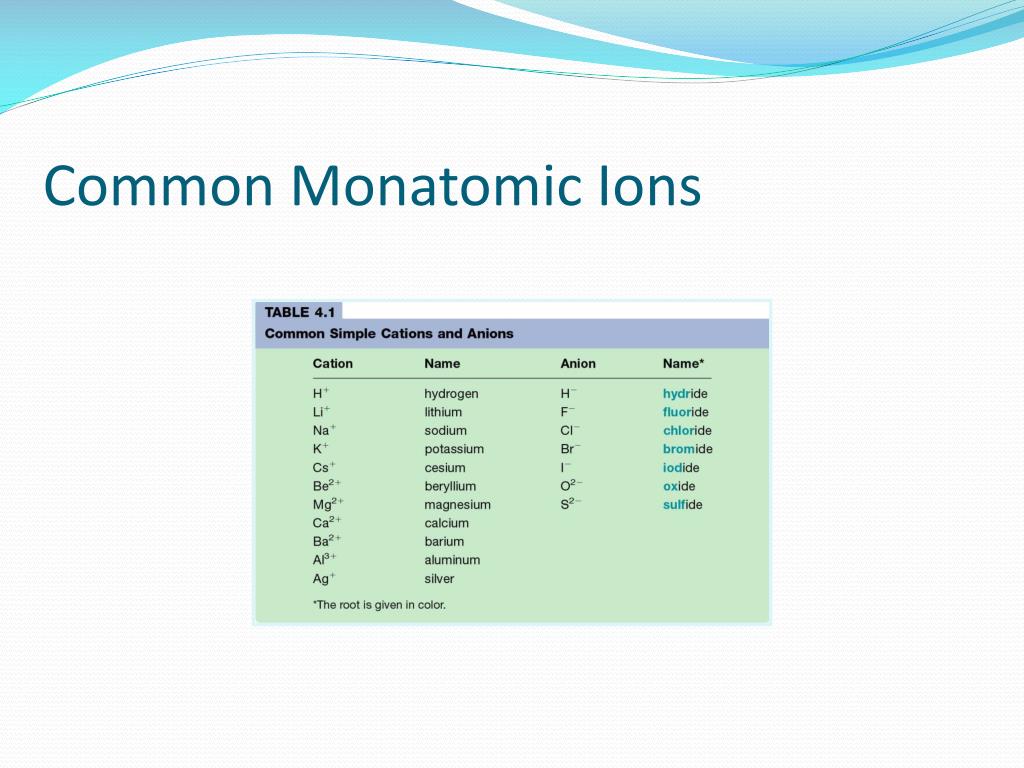
Thus, the most stable ion of cesium is the 1+ ion, or Cs +. Cesium is in group 1 so it has 1 valence electron which it want to lose. The light reflected from the surface of a pool of water is observed through a. For stable elements, there is usually a variety of stable isotopes. University Professor with 10+ years Tutoring Experience. Neon is a very inert element, however, it has been reported to form a compound. The covalent radius of Cl 2 is half the distance between the two chlorine atoms in a single molecule of Cl 2. What is the most stable monatomic ion formed from caesium Verified answer. Ionic radius, r ion, is the radius of a monatomic ion in an ionic crystal. (d) This is a depiction of covalent versus van der Waals radii of chlorine. Monatomic ions are formed by the gain or loss of electrons to the valence shell (the outer-most electron shell) in an atom. What is the most stable monatomic ion formed from fluorine How many pairs of valence electrons do the bromine atoms have in the following molecules and ions. (c) The van der Waals atomic radius, r vdW, is half the distance between the nuclei of two like atoms, such as argon, that are closely packed but not bonded. S8) using the newly-identified most stable NHC-cation configurations, different results. What is the most stable monatomic ion formed from caesium Answers Answer 1 Answer: The most stable ion of cesium is the 1+ ion, or Cs+. The dynamics of collisional processes between Mg atoms and caesium ions is. (b) The metallic atomic radius, r met, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. Iron, silver, nickel), whilst most other nonmetals typically form anions (e. (a) The covalent atomic radius, r cov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. Trending Questions Is a piece of ice melting and reacting with sodium a physical or chemical change? Which metal is preserved in kerosine? The hydrogen atom has one what and one what? What are the 4 organic compounds? What does the two bold dot symbol indicate on a weather map? NaCI is it a mixture or compound? What is the famous hurricane? In the formula for sodium phosphate Na3PO4 how many moles of sodium are represented? Which type of moving water provides the best environment for organisms that decompose dead organic matter? Why don't tornadoes hit big cities? What term describes specific areas where the earths plates move against each other and produce earthquakes? What are the flattest places on earth? Why would the Kelvin scale not be a practical scale of temperature for everyday use? Which group 5A element forms the most basic oxide? The process that water goes through as it changes from one state to another? What is the Mass in Grams of 1.\): Definitions of the Atomic Radius. What is the energy of a bond formed between a potassium (K+) cation and an iodide (I-) anion The ionic radii of K+ and I-, are 152 pm and 206 pm, respectively. 1 Common Monoatomic Ions H-hydride Na + sodium H+ hydrogen Li + lithium fluorideF- Cs + caesium K+ potassium Ag + silver Cl - chloride Br - bromide I-iodide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
